JH-RE-06
Select A Tab:
A novel inhibitor of mutagenic translesion synthesis (TLS), disrupting mutagenic TLS by preventing recruitment of mutagenic POL ζ, targets a nearly featureless surface of REV1 that interacts with the REV7 subunit of POL ζ, inducing REV1 dimerization to block the REV1-REV7 interaction.
| Technical Data | |
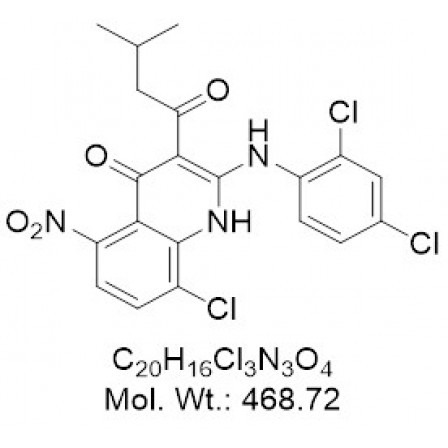
| Chemical Formula | C20H16Cl3N3O4 |
| CAS Number | 1361227-90-8 |
| Molecular Weight | 468.72 |
| Solubility | DMSO |
| IUPAC/Chemical Name | 8-chloro-2-((2,4-dichlorophenyl)amino)-3-(3-methylbutanoyl)-5-nitroquinolin-4(1H)-one |
| Purity | 98% by HPLC |
| Storage | 0°C (short term), -20°C (long term), desiccated |
| InChI Key | LRTXIQCBQIKIOH-UHFFFAOYSA-N |
| InChl Code | InChI=1S/C20H16Cl3N3O4/c1-9(2)7-15(27)17-19(28)16-14(26(29)30)6-4-11(22)18(16)25-20(17)24-13-5-3-10(21)8-12(13)23/h3-6,8-9H,7H2,1-2H3,(H2,24,25,28) |
| Smiles | ClC1=C(NC(NC2=CC=C(Cl)C=C2Cl)=C(C(CC(C)C)=O)C3=O)C3=C([N+]([O-])=O)C=C1 |